Eternal Jellies in the World Ocean

Abundant moon jellies in the Baltic shallows
I have lived much of my adult life here in landlocked Winnipeg, but I grew up much closer to the sea and spent many summers in my 20s doing fieldwork beside the mercurial Atlantic and the more peaceable Baltic. It is strange to consider that I now live in a place that may be almost as far from the sea as anywhere in North America, yet most days I spend hours contemplating marine organisms present and past, trying to understand the environments of ancient shorelines and long-lost salt seas. I often feel the emotional tide of the ocean, and every year I try to spend days or weeks absorbed by its sights, sounds, and smells. Sometimes I collect fossils beside the cold and often fog-bound Hudson Bay, but I have also had many days beachcombing on both sides of the Atlantic, along the Baltic, Mediterranean, and Caribbean, and on the east and west shores of the Pacific.
For the past several years, I have been trying to make sense of 445 million year-old fossil jellyfish that we discovered in central Manitoba (1). These are, effectively, jellyfish that were washed up on an ancient shore and buried in fine sediment before they had a chance to rot away completely. They are strange fossils, and difficult to understand, so each time we go to the seashore, my patient family has to put up with my constant searching for beached modern jellyfish to examine and photograph (“Daddy’s looking for jellyfish. Again!“).
Ordovician fossil jellyfish from central Manitoba (The Manitoba Museum, specimen I-4056A)
Some of these modern jellies are remarkably widespread organisms. I have looked at examples of the moon jelly Aurelia in Florida, in the Baltic, and in the Pacific, and they are all very similar. They may differ in how much “jelly” they have, or in what colour their gonads are, and they apparently represent a few species (the experts don’t seem to really agree), but nonetheless, this is obviously a type of organism that can be found in seas and oceans almost everywhere. Similarly, I have examined the lion’s mane jellyfish Cyanea capillata on the coasts of Scotland, Prince Edward Island, and Hudson Bay. How many other kinds of organisms are this widespread? There must be billions of jellyfish in this world, yet all of them belong to a remarkably small number of species – it is estimated that there are only about 200 species of “true jellyfish” (Scyphozoa), plus 20 species of box jellies (Cubozoa), and 1000-1500 species of the hydrozoan jellyfish (hydromedusae; many of these are minute, almost microscopic in size). By way of comparison, there are hundreds of thousands of species of mosquitoes, gnats, midges, and their relatives; jellyfish diversity is very different from that of the land-dwelling animals with which we are more familiar.

Dead moon jellies, Island of Saaremaa, Estonia

Lion's mane jelly, northeast tip of Prince Edward Island
How can it be that the diversity of jellyfish is so relatively meagre, when the oceans are far larger in area than the land, and when jellyfish are widespread and abundant through much of this three-dimensional medium? Much of the answer, as far as I can determine, lies in basic differences between land and water environments. When I stand on the shore of the Baltic, or the shore of the Pacific, I am effectively on the edge of the same body of water. These seas and oceans, even if they are on opposite sides of the planet, are linked by liquid, as indeed are all oceanic bodies of water. This is most obvious when the Earth is looked at from the south polar perspective, since around Antarctica the Atlantic, Pacific, and Indian oceans all flow together to form the “Great Southern Ocean.” But follow the tentacles of that ocean northward, and you will see that it links into the Caribbean, the South China Sea, the North Sea, the Bering Sea, even dear old Hudson Bay. To animals that live within the water column, these water bodies are all one, and the animals move around the world as the currents, water conditions, and food resources allow.
This situation has been similar through most of geological time. Four hundred million years ago, the land of the world was divided into continents that would be completely unfamiliar to our eyes, but they were surrounded and partly covered by that same liquid environment. In the intervening millennia, continents have risen and fallen, land has come and gone, but the ocean has changed very gradually, with only a few jarring knocks to trouble its otherwise placid existence.

The Great Southern Ocean, as seen from above the South Pole (Wikimedia Commons)
Since the ocean is a liquid, not a solid, its shape is not so material to the organisms that make it their home, particularly those that spend their lives without touching land or the seafloor. The great majority of these life forms fit within the broad definition of plankton, which basically includes all those things that drift with the currents. Animal plankton, or zooplankton, are immensely varied: crustaceans, salps, polychaete worms, gastropods, larval fish, and numerous other forms. Many of these are microscopic, and most jellyfish are at the “macro” end of the zooplankton scale. Since the really big true jellies are among the largest of the plankton, they are the easiest for the casual observer (such as myself) to see, and, perhaps, to understand.

Crystal jelly Aequorea (a big hydromedusan), Pacific coast of Canada (photo: Gavin Hanke)
It was recognized long ago that the evolution patterns for the plankton are quite different from those of organisms that live attached to the seafloor, which are different again from land animals. Many plankton species are extremely long-lived, as are the groups to which they belong (2). Since many zooplankton (such as my familiar jellyfish) are geographically widespread and exist in unimagineably huge numbers, they may be protected from most of the phenomena that cause other things to become extinct. But their numbers and distribution are themselves related to what is called the “paradox of the plankton.” The basic idea is that zooplankton, living in such immense and widespread populations, surrounded and fed by water, are in a situation where physical conditions affect them much more than any competition between species. The paradox of the plankton uses this to explain the generally high diversity of plankton, but a lack of direct competition could also potentially explain the relatively low diversity of jellyfish. New species arise where selective forces are driving evolutionary change, often on small and separated populations; the huge populations, continuous distribution, and apparently low competition among jellyfish make it unlikely that new species would arise frequently.
Some groups of plankton have hard skeletons and have left good fossil records in sedimentary rocks all over the world. Others, such as jellyfish, lack hard parts, and are remarkably rare and sporadic as fossils. Together with my colleague Whitey Hagadorn of Amherst College, I have been working on a review of the fossil record of jellyfish (3). There are only a few good jellyfish sites in the entire fossil record, but several recent papers have described ancient forms that are remarkably similar to those we can see today (e.g., 4, 5). Hundreds of millions of years ago, moon jellies and box jellies and many other forms were washing up on shorelines, just as they do today.

Modern moon jellies (Aurelia) from the southern end of Vancouver Island

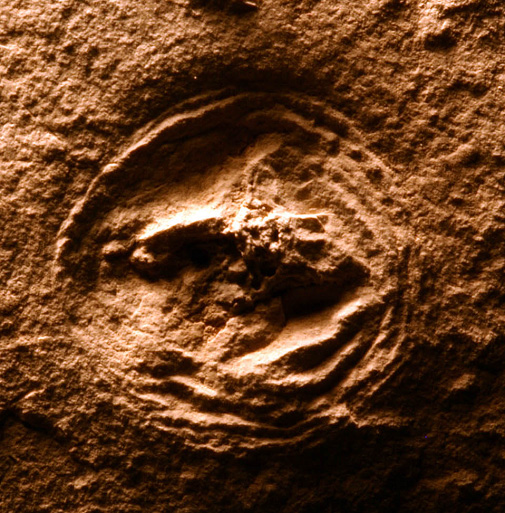
This fossil jelly, similar to a moon jelly, was discovered in a schoolyard in Winnipeg. Its age is uncertain, but the rock in which it occurs is consistent with an Ordovician age, about 450 million years old (The Manitoba Museum, specimen I-2555).
It appears that, once planktonic organisms find their niches, it may take some sort of cataclysmic event to displace them. They seem to be immune to the sorts of ordinary “day-to-day” factors that caused the extinction of mere mortals such as the dodo or mammoth, and that threaten so many other creatures today. It is still likely, however, that mass extinction events will affect them. Many groups of skeletonized zooplankton were severely affected by the end Cretaceous extinction, the same catastrophe that saw the demise of the dinosaurs and pterosaurs. The jellyfish fossil record is so sporadic that we can’t say whether that event made them miss their dinners, but I suspect that at least some of them must have been pushed onto the “extinct” side of the ledger.
I plan to write more pieces about fossil jellyfish, and about modern jellyfish blooms, but today in snowy Winnipeg I feel much more compelled by thoughts of their relationship to the ocean itself, in all of its endless and trackless wonder.
References
(1) Young, G. A., Rudkin, D. M., Dobrzanski, E. P., Robson, S. P., & Nowlan, G. S. 2007. Exceptionally preserved Late Ordovician biotas from Manitoba, Canada. – Geology 35, 10: 883-886; Boulder.
(2) Rigby, S. & Milsom, C. V. 2000. Origin, evolution, and diversification of zooplankton. – Annual Reviews of Ecology and Systematics, 31: 293-313; Palo Alto.
(3) Young, G.A. and Hagadorn, J.W. Accepted for Publication. The fossil record of cnidarian medusae. Proceedings, X International Symposium on Fossil Cnidaria and Porifera, St. Petersburg.
(4) Gaillard, C., Goy, J., Bernier, P., Bourseau, J.P., Gall, J.C., Barale, G., Buffetaut, E., & Wenz, S. 2006. New jellyfish taxa from the Upper Jurassic lithographic limestone of Cerin (France): taphonomy and ecology. – Palaeontology, 49, 6: 1287-1302; Oxford.
(5) Cartwright, P., Halgedahl, S. L., Hendricks, J. R., Jarrard, R. D., Marques, A. C., Collins, A. G., & Lieberman, B. S. 2007. Exceptionally preserved jellyfishes from the Middle Cambrian. – PLoS ONE, 2: e1121 (online journal).


I discovered what looks like a jelly fish (a bit like the modern Aequorea you picture above). These are Ordovician rocks from the Ottawa Group- Bobcagen Formation…
Any idea what they might be
Size- approx. cantolope size with a radial sun-like spread of ‘fingers’ around periphery of circular centre…
Hi Stephanie, I suspect that they are fluid escape structures. Those often develop a form quite similar to Aequorea, especially if they are in fine-grained, algal-laminated sediments. Still, there is the chance that they could be jellyfish; can you send photos to my e-mail?